Return
to research document list...
|
Int. J. Immunotherapy X1(1) 23-28 (1995)
IMMUNOMODULATORY AND ANTICANCER EFFECTS OF ACTIVE HEMICELLULOSE COMPOUND (AHCC) GHONEUM M., 1* WIMBLEY.M., 2 SALEM F., 3 MCKLAIN A., 1 ATTALLAH N., 2 GILL G.1
*Address for correspondence: Drew University of Medicine and Science, Department of Otolaryngology, 1621 East 120th Street, Los Angeles, CA 90059 USA. Summary: The effects of therapy with active hemicellulose compound (AHCC) were examined in 11 cancer patients. AHCC is a myecelic extract of basidiomycota originating from hybrid mushrooms. Significant anticancer activity by AHCC was observed with advanced malignancies in patients given 3 g of AHCC daily. The percentages of patients with complete remission were as follows: (i) prostatic, 213 (66%). PSA level <0.2; (ii) ovarian, 213 (66%). CA 125 < 35; (iii) multiple myeloma, _ (50%), BJP <5; (iv) breast, 1/3 complete remission and 2 partial. Two mechanisms by which AHCC exerts its effect were investigated. The first was natural killer (NK) immunomodulation. Patients demonstrated a low base level of NK activity (18.8%), which was significantly enhanced by AHCC at 2 weeks (2.5 fold), and was maintained at a high level. The second was direct anticancer properties. In vitro studies showed that AHCC possesses suppressive effects on tumour cell growth. AHCC (1 mg/ml) cultured with K562 and Raji tumour cells caused 21% and 43% decrease in cell counts, respectively, as compared to control untreated cells. It is concluded that the high augmentory effect of AHCC and the absence of notable side-effects make AHCC a promising immunotherapeutic agent for the treatment of cancer patients.
Introduction
The theory of immune surveillance postulates that immune effectors can recognize and destroy spontaneously arising malignant tumour cells. Tumours may develop when transformed cells escape immunological host defense mechanisms. The increased incidence of spontaneous tumours in immunosuppressed individuals, as well as those with congenital or acquired immunodeficiencies, indicates that the immune system can provide a major mechanism for host resistance against cancer and infectious diseases. The idea of immune surveillance has gained renewed interest with the discovery of natural killer (NK) cells (1 — 4). Several attempts have been made to stimulate the immune system for tumour fighting. Several biological response modifiers (BRMs) have been developed, such as: Poly 1:C, PCG, Corynebacterium parvum, interferon and interleukin-2 (IL-2). However, the clinical use of these BRMs is limited because of their severe side effects. For example IL-2 treatment may cause kidney failure, capillary leaking syndrome, vomiting, nausea and many other side-effects, in addition to the high cost of treatment which may reach $100,000 for single IL-2 therapy for hospitalized patients. The authors were therefore interested in examining a new BRM which enhances NK activity without causing side-effects. It is interesting to note that active hemicellulose compound (AHCC) possess these two features. Eleven cancer patients with different types of advanced malignancies participated in the study. Results showed enhancement of NK activity after treatment with AHCC, which resulted in complete or partial remission of the disease. Patients, material and methods Patients Eleven randomly selected patients with different types of advanced malignancies were included in the study. Patients were treated with conventional therapies such as surgery, chemotherapy and radiation. All patients had a pathological report confirming a diagnosis indicating the type and stage of cancer. The type of chemotherapy was obtained from medical records. There were several types of cancers in the study, as follows: prostatic (3), ovarian (3), multiple myeloma (2) and breast (3). Patients ranged in age from 36 to 65 years with a mean age of 53 (See Table 1.) Table 1 Histological Diagnosis of cancer patients and type of conventional therapy
*Means treatment is continued.
Material Complete medium (CM). It consisted of RPMI-1640 supplemented with 5% fetal calf serum (FCS), 100 U penicillin and 100 µg/ml streptomycin Active hemicellulose compound (AHCC). AHCC is a mycelia extract of the mushroom basidiomycota. Basidiomycota originated from the hybridization of several types of mushrooms. It contains polysaccharides (b -1,3 glucan and activated hemicellulose) and glycoproteins. Methods Treatment protocol. Patients were given AHCC (3 g/day) daily by mouth. Tumour-associated antigens (TAA). TAA for each type of malignancy was measured prior to AHCC treatment and at 3 to 40 week intervals. (See Table II).
Table II Tumour-associated antigen (TAA) for different types of malignancies.
Preparation of mononuclear cells. Blood (10ml) was drawn from each patient before treatment, at 2 weeks post treatment and then at monthly intervals. Mononuclear cells were separated from heparinized peripheral venous blood by Ficoll-Hypaque density-gradient centrifugation. Cells were washed three times with Hanks balanced salt solution and resuspended to a concentration of 1 x 107/ml. This suspension was used to examine NK activity. NK cell cylotoxicity assay. The standard 51Cr-release assay was employed. Briefly, 1 x 104 51Cr labeled K562 target cells (New England Nuclear Corporation) in 0.1 ml CM were added per well in microlitre plates. Effector cells were pipetted into quadruplicate wells to give an effector:target cell ratio of 100:1. These cells were allowed to interact at 37°C for 4 h in an atmosphere of 5% CO2/95% air. 51Cr release was determined by centrifuging the plates at 1000 g for 5 min and harvesting 0.1 ml of the culture supernatant for counting in a gamma counter. Total release was determined by adding 100 µl of 1.0% Trition X-100, and spontaneous release was determined by adding labeled target cells alone in CM. The percent 51CR-release was determined from the experimental (Rc), spontaneous (Rs) and total (Rl) release by the following formula: % 51Cr-release =
In vitro studies. Two different tumour cell-lines were used in the study: K562 (an erythroleukaemia cell-line) and Raji (a Burkett cell lymphona). Cell-lines were maintained via passage three times a week. Cells were adjusted to 1 x 105cells/ml and cultured in CM containing 2.5% FCS in the presence or absence of AHCC. AHCC was dissolved in PBS at concentrations of 0.5 and l mg/ml. A cell count was carried out daily for 6 days using a haemocylometer. Cell viability was examined by trypan blue stain. Statistical analysis. A two-tailed Student’s t-teat was used to determine the degree of significance between NK cell activities before and after treatment with AHCC.
Results Tumour-associated antigens (TAA) The level of TAA for each type of malignancy pre/post treatment with AHCC is presented in Table III. Prostatic cancer. PSA was the marker evaluated in prostatic cancer. All patients demonstrated a significant decline in the level of PSA. There was a rapid decrease of the PSA level in two patients. They then reached normal levels of PSA at 1 to 2 months post treatment. Ovarian cancer. A significant decline in the level of CA 125 was observed in two out of three patients with ovarian carcinoma. The rapid decrease in the level of CA 125 occurred at 1 month and continued to decline until it reached normal value (35 Units), 3 to 4 months post treatment. Multiple myeloma. Two patients participated in the study. The first patient had a BJP of 150. After AHCC treatment, the BJP level declined to 10 and <5 at 11 and 17 months, respectively. The second patient was followed with abnormal protein. The baseline was 3.2 prior to treatment and decreased to 2.6 in 3 months. Breast cancer. The level of CA 15-3 in cancer patients was low and did not change after AHCC therapy.
Table III levels of tumour=associated antigens (TAA) post treatment with AHCC
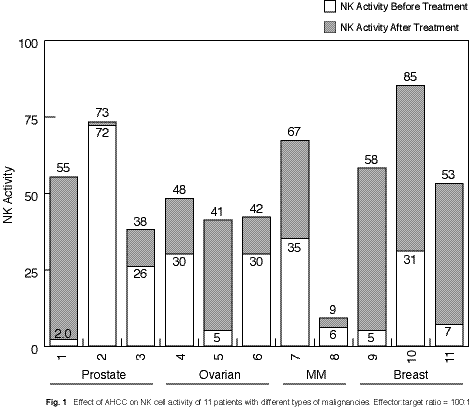
NK cell activity As shown in Fig. 1, patients demonstrated an overall low level of basal NK activity (18.8%), that was significantly enhanced by AHCC at 2 weeks (2.5-fold), and was maintained at a high level. Patients showed differences in susceptibility toward the immunomodulatory function of AHCC. Nine out of eleven patients had an increase in their NK activity after treatment with AHCC. Direct anticancer activityAn in vitro study showed that AHCC possesses direct anticancer activity against the tumour cell-lines K562 and Raji. The effect was dose dependent. AHCC suppresses the growth of both cell-lines, but Raji cells are more sensitive than K652. AHCC at a concentration of 1 mg/ml caused 21% and 43% reduction in the growth of K564 and Raji cells, respectively (Table IV). Table IV Effect of AHCC on tumour cell growth in vitro* Concentration of AHCC (mg/ml)
*Cells were examined 6 days after treatment. FCS concentration 2.5%. Data represent mean ± s.d. of three experiments. b Served as control.
Discussion Several BRM’s have been the object of great interest because of their immunomodulatory functions and their potential value in tumour therapy. Data from the present study demonstrates that AHCC is a promising anticancer agent, as manifested by a significant decline of TAA in 8 out of 11 patients with different types of malignancies. Levels of prostatic PSA and ovarian CA 125 decreased markedly as early as 1 to 2 months, and reached the normal level within 1 to 4 months. On the other hand, levels of BJP and ab. protein declined significantly but took longer. With respect to CA 15-3, patients had normal levels of this marker and it did not change appreciably after AHCC therapy. The mechanisms by which AHCC exerts its effect were examined. First the NK cell immunomodulatory functions of AHCC were evaluated. NK cells are considered to be the first line of defense against cancer (1-4). Results showed that AHCC is a potent immunomodulator. Nine out of eleven patients demonstrated a marked increase in NK activity as early as 2 weeks after treatment with AHCC. The activity was maintained at a high level. Earlier studies in the author’s laboratory showed enchancement of murine NK cell activity by AHCC using old mice as a model. AHCC injected i.p. caused a 3 to 4-fold increase of peritoneal NK activity at 3 to 14 days (5), and experiments carried out with healthy control subjects revealed a significant increase of PBL-NK activity after administration of AHCC (3g/d) for 2 weeks (6). The observed increase in NK cell activity was due to an increase in NK cell granularity, as well as the binding capacity of NK cells to their tumour cell targets (7). Another set of experiments was carried out to investigate possible direct anticancer activity by AHCC against tumour cell-lines in vitro. Two tumour cell-lines were used in the study: K562 (an erythroleukaemia cell-line) and Raji (a Burkitt cell lymphoma) of human origin. Results showed that AHCC inhibits tumour cell growth in a dose-dependent manner. Both tumour cell-lines were sensitive towards AHCC toxicity but Raji cells were more sensitive than K562. Conclusions AHCC is a new anticancer agent that does not have any known side-effects. This aspect provides an additional advantage of AHCC over the other BRMs. The mechanism(s) by which AHCC exerts its anticancer activity may be through: (i) NK cell immunomodulation; (ii) direct anticancer activity. Further studies are needed to investigate the effect of AHCC in multiple clinical trails.
Acknowlegements AHCC was offered by Daiwa Pharmaceutical Co., Tokyo 151, Japan. Research was supported by NIH/MBRS (BR-08149). The authors appreciate the technical support of Ms. Marcia Pugh and Miss Horuno Kobayashi. References
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||